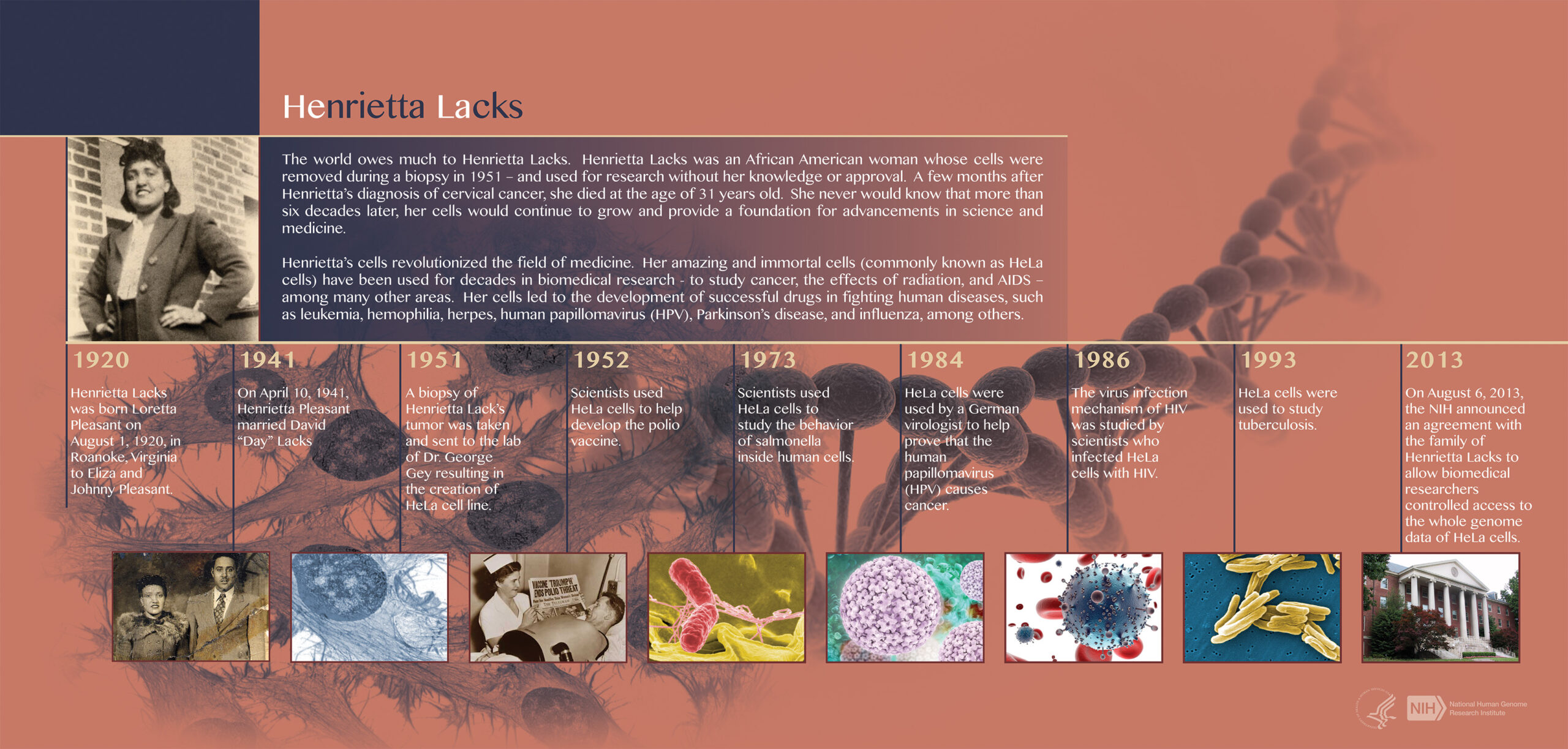
Legacy of Henrietta Lacks

Courtesy of March of Dimes
Henrietta Lacks was a poor, Black tobacco farmer whose cells were taken without her permission during her treatment for cervical cancer in 1951, decades before federal law prohibited taking biological samples without consent. These cells have been used to culture an endless supply of cells for medical research and have been the subject of thousands of scientific papers. Lack’s family members, who have not benefited financially from her legacy, did not know for decades that her cells were being used. After the full genome was posted publicly, concerns over privacy prompted discussion about the ethics surrounding the use of genetic material.
In 1952, the Tuskegee Institute—which had had nothing to do with obtaining Henrietta Lacks’ tissue—was selected by The Infantile Paralysis Foundation to carry out the HeLa cell culture project. As of August 2013, two members of the Lacks family sit on the six-member committee that regulates access to the HeLa genetic code by research projects funded by the National Institutes of Health.
In February 2026, the second of many lawsuits brought by the Lacks family against biotech and medical companies profiting off their use of Henrietta’s cells ended when the Swiss pharmaceutical company Novartis settled with the Lackses for an undisclosed amount.
Henrietta Lacks’ life and the life of her “HeLa” cells:
Further Reading
Beskow, L. M. (2016). Lessons from HeLa Cells: The Ethics and Policy of Biospecimens. Annual Review of Genomics and Human Genetics, 17, 395–417.
Garrison, N. A. (2015). Considerations for Returning Research Results to Culturally Diverse Participants and Families of Decedents. The Journal of Law, Medicine & Ethics : A Journal of the American Society of Law, Medicine & Ethics, 43(3), 569–575.